Summary
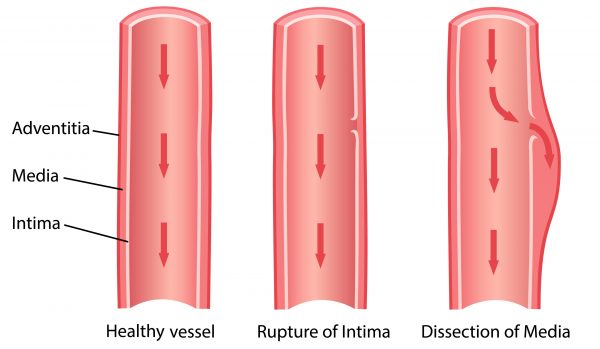
The aorta is a large blood vessel divided into the ascending aorta rising from the heart, the aortic arch where the aorta changes direction; and the descending aorta ending with iliac bifurcation. The aortic wall consists of three layers: intima, media, and adventitia. One of the injuries affecting the innermost layer of the aorta is aortic dissection (AD), i.e. a tear in the intima that enables blood flow between the layers of the aortic wall, forcing the layers further apart, and thus creating a true lumen (TL) and a false lumen (FL). In case the blood-filled channel ruptures through the outside of the aortic wall, AD is often fatal. It is a relatively uncommon disease; the prevalence is approximately 30 cases per million individuals per year. It is also challenging to diagnose and has high mortality rate.
There is still no clear clinical consensus whether medical or endovascular treatment gives better results for uncomplicated type B aortic dissection. Additionally, a difficult decision to make is at which point to stop with the medical treatment and intervene, either surgically or endovascularly.
In order to assist clinical decision making, a trend toward patient-specific computational modelling has emerged in recent years.
Stages of aortic dissection.
Goal
The aim of the project is the development of a comprehensive biochemomechanical FSG model of AD evolution that will increase our understanding of biochemomechanical processes occurring within AD. It will also allow us to theorize about optimal treatment options and predict rupture. Future application of the model should be able to predict the possible response of an artery to a graft implementation or how pharmaceuticals may affect AD progression. This prognosis tool would help clinicians to optimize treatment, possibly lower the number of life-threatening events and reduce the number of unnecessary costly surgical operations.
This goal will be achieved through 3 main objectives:
- Collecting patient data necessary for the realization of the project goal.
Appropriate patients with an uncomplicated Type B aortic dissection will be selected. The collected data will include information about patient anamnesis, AD morphology, blood tests, and laboratory data. MRI imaging will be performed in order to determine blood flow. Data will be collected at Sestre milosrdnice University Hospital Center and from the IRAD database.
- The development of a fluid-solid-growth (FSG) framework.
The existing Growth&Remodelling (G&R) model will be adapted to the thoracic aortic wall structure. The model will be verified on the known clinical observations (such as changes in blood flow or pressure) and further adapted to model development of the aortic dissection. It will account for changes in mass, structure, and mechanical behaviour of the wall during AD evolution. Simultaneously, an FSI model will be developed. The aortic wall will not be simplified as rigid. The model will be used to determine crucial hemodynamic parameters such as the pressure distributions within the true and the false lumen, i.e. loads used in the G&R model. Since thrombus developing within the false lumen is likely the key factor in determining the rupture risk focus will also be put on dealing with a G&R model of thrombus development. Finally, all the models developed in the previous three WPs will be combined into one FSG model of AD.
- The use of the developed FSG model
from objective 2 over several years or decades to predict an outcome and potential rupture risk of uncomplicated TBAD thus reducing the number of life-threatening events and unnecessary costly surgical operations.